Background
Multi-regional clinical trials (MRCTs) are increasingly used in global drug development to allow simultaneous regulatory submissions across multiple regions. A key requirement for regional approval — particularly in Japan under the Japanese MHLW guidelines — is the demonstration of regional consistency: evidence that the treatment effect observed in a specific region (e.g., Japan) is consistent with the overall trial result.
Two widely used consistency evaluation methods, originally proposed under the Japanese guidelines, are:
- Method 1 (Effect Retention Approach): Evaluates whether Region 1 retains at least a fraction of the overall treatment effect.
- Method 2 (Simultaneous Positivity Approach): Evaluates whether all regional estimates simultaneously show a positive effect in the direction of benefit.
These methods were originally developed for two-arm randomised controlled trials. However, single-arm trials are now common in oncology and rare disease settings, where historical control comparisons are standard. The SingleArmMRCT package extends Method 1 and Method 2 to the single-arm setting, in which the treatment effect is defined relative to a pre-specified historical control value.
Regional Consistency Probability
The Regional Consistency Probability (RCP) is defined as the probability that a consistency criterion is satisfied, evaluated under the assumed true parameter values at the trial design stage. A trial design is said to have adequate regional consistency if the RCP exceeds a pre-specified target (commonly 0.80).
Method 1: Effect Retention Approach
Let denote the endpoint parameter for a given endpoint (e.g., mean, proportion, rate). Method 1 requires that Region 1 retains at least a fraction of the overall treatment effect:
where is the treatment effect estimate for Region 1, is the overall pooled estimate, is the null (historical control) value, and is the pre-specified retention threshold (typically ).
The consistency condition can be rewritten as , where:
with being the regional allocation fraction and the pooled estimate for regions combined. Under the assumption of homogeneous treatment effects across regions, follows a normal distribution with mean and a variance that depends on the endpoint type, yielding a closed-form expression for , where is the treatment effect.
For endpoints where a smaller value indicates benefit (e.g., hazard ratio, rate ratio), the inequality direction is reversed. See the endpoint-specific vignettes for exact formulae.
Method 2: Simultaneous Positivity Approach
Method 2 requires that all regional estimates simultaneously demonstrate a positive effect. For endpoints where a larger value indicates benefit (continuous, binary, milestone survival, RMST):
For endpoints where a smaller value indicates benefit (hazard ratio, rate ratio):
Because regional estimators are independent across regions, factorises as:
Package Structure
The package provides a pair of functions for each of six endpoint types.
| Endpoint | Calculation function | Plot function |
|---|---|---|
| Continuous | rcp1armContinuous() | plot_rcp1armContinuous() |
| Binary | rcp1armBinary() | plot_rcp1armBinary() |
| Count (negative binomial) | rcp1armCount() | plot_rcp1armCount() |
| Time-to-event (hazard ratio) | rcp1armHazardRatio() | plot_rcp1armHazardRatio() |
| Milestone survival | rcp1armMilestoneSurvival() | plot_rcp1armMilestoneSurvival() |
| Restricted mean survival time (RMST) | rcp1armRMST() | plot_rcp1armRMST() |
Each calculation function supports two approaches:
-
"formula": Closed-form or semi-analytical solution based on normal approximation. Computationally fast and, for binary and count endpoints, exact. -
"simulation": Monte Carlo simulation. Serves as an independent numerical check of the formula results.
Common Parameters
All six calculation functions share the following parameters.
| Parameter | Type | Default | Description |
|---|---|---|---|
Nj |
integer vector | — | Sample sizes for each region; length equals the number of regions |
PI |
numeric | 0.5 |
Effect retention threshold for Method 1; must be in |
approach |
character | "formula" |
Calculation approach: "formula" or
"simulation"
|
nsim |
integer | 10000 |
Number of Monte Carlo iterations; used only when
approach = "simulation"
|
seed |
integer | 1 |
Random seed for reproducibility; used only when
approach = "simulation"
|
Time-to-event endpoints (hazard ratio, milestone survival, RMST) additionally require the following trial design parameters.
| Parameter | Type | Default | Description |
|---|---|---|---|
t_a |
numeric | — | Accrual period: duration over which patients are uniformly enrolled |
t_f |
numeric | — | Follow-up period: additional observation time after accrual closes; total study duration is |
lambda_dropout |
numeric or NULL
|
NULL |
Exponential dropout hazard rate; NULL
assumes no dropout |
Quick Start Example
The following example computes RCP for a continuous endpoint with the setting below:
| Parameter | Value |
|---|---|
| Total sample size | ( regions) |
| Region 1 allocation | () |
| True mean | |
| Historical control mean | (mean difference ) |
| Standard deviation | |
| Retention threshold |
Closed-form solution
result_formula <- rcp1armContinuous(
mu = 0.5,
mu0 = 0.1,
sd = 1,
Nj = c(10, 90),
PI = 0.5,
approach = "formula"
)
print(result_formula)
#>
#> Regional Consistency Probability for Single-Arm MRCT
#> Endpoint : Continuous
#>
#> Approach : Closed-Form Solution
#> Target Mean : mu = 0.5000
#> Null Mean : mu0 = 0.1000
#> Std. Dev. : sd = 1.0000
#> Sample Size : Nj = (10, 90)
#> Total Size : N = 100
#> Threshold : PI = 0.5000
#>
#> Consistency Probabilities:
#> Method 1 (Region 1 vs Overall) : 0.7446
#> Method 2 (All Regions > mu0) : 0.8970Monte Carlo simulation
result_sim <- rcp1armContinuous(
mu = 0.5,
mu0 = 0.1,
sd = 1,
Nj = c(10, 90),
PI = 0.5,
approach = "simulation",
nsim = 10000,
seed = 1
)
print(result_sim)
#>
#> Regional Consistency Probability for Single-Arm MRCT
#> Endpoint : Continuous
#>
#> Approach : Simulation-Based (nsim = 10000)
#> Target Mean : mu = 0.5000
#> Null Mean : mu0 = 0.1000
#> Std. Dev. : sd = 1.0000
#> Sample Size : Nj = (10, 90)
#> Total Size : N = 100
#> Threshold : PI = 0.5000
#>
#> Consistency Probabilities:
#> Method 1 (Region 1 vs Overall) : 0.7421
#> Method 2 (All Regions > mu0) : 0.8922The closed-form and simulation results are in close agreement. The
small difference is attributable to Monte Carlo sampling variation and
diminishes as nsim increases.
Visualisation
Each endpoint type has a corresponding plot_rcp1arm*()
function. These functions display RCP as a function of the regional
allocation proportion
,
with separate facets for different total sample sizes
.
Both Method 1 (blue) and Method 2 (yellow) are shown, with solid lines
for the formula approach and dashed lines for simulation. The horizontal
grey dashed line marks the commonly used design target of RCP
.
The base_size argument controls font size: use the
default (base_size = 28) for presentation slides, and a
smaller value (e.g., base_size = 11) for documents and
vignettes.
plot_rcp1armContinuous(
mu = 0.5,
mu0 = 0.1,
sd = 1,
PI = 0.5,
N_vec = c(20, 40, 100),
J = 3,
nsim = 5000,
seed = 1,
base_size = 11
)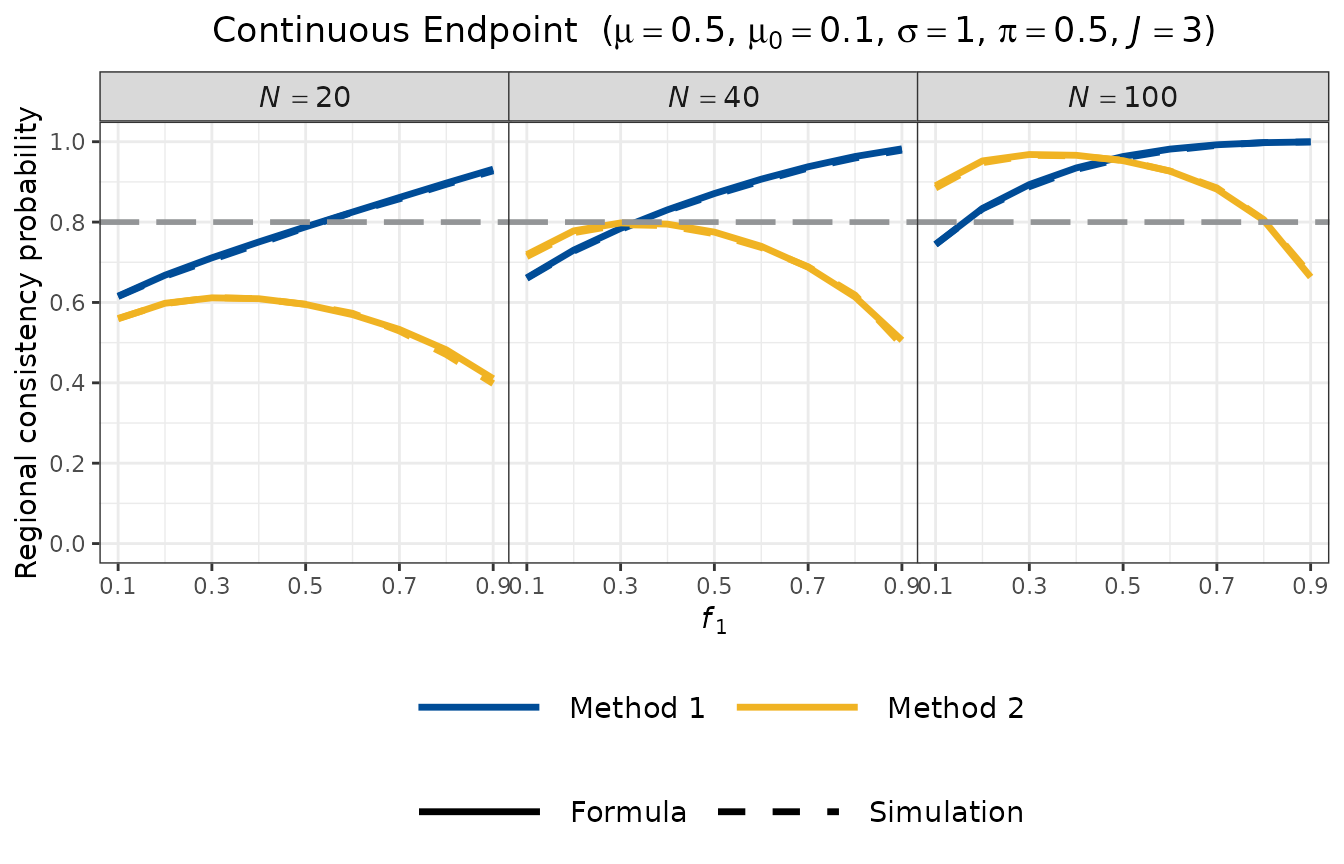
Several features are evident from the plot:
- Method 1 (blue) increases with : as Region 1 becomes larger, its estimator becomes more precise, making the retention condition easier to satisfy.
- Method 2 (yellow) is maximised when all regions have equal allocation , and decreases as deviates from this balance, because unequal allocation reduces the marginal probability for the smaller regions.
- Both RCP values increase with total sample size , as expected.
- The formula (solid) and simulation (dashed) curves are closely aligned, confirming the accuracy of the normal approximation.
Further Reading
For endpoint-specific statistical models, derivations, and worked examples, see the companion vignettes:
- Non-survival endpoints: continuous, binary, and count (negative binomial) endpoints.
- Survival endpoints: hazard ratio, milestone survival probability, and RMST endpoints.
References
Hayashi N, Itoh Y (2017). A re-examination of Japanese sample size calculation for multi-regional clinical trial evaluating survival endpoint. Japanese Journal of Biometrics, 38(2): 79–92. https://doi.org/10.5691/jjb.38.79
Homma G (2024). Cautionary note on regional consistency evaluation in multiregional clinical trials with binary outcomes. Pharmaceutical Statistics, 23(3):385–398. https://doi.org/10.1002/pst.2358
Wu J (2015). Sample size calculation for the one-sample log-rank test. Pharmaceutical Statistics, 14(1): 26–33. https://doi.org/10.1002/pst.1654
